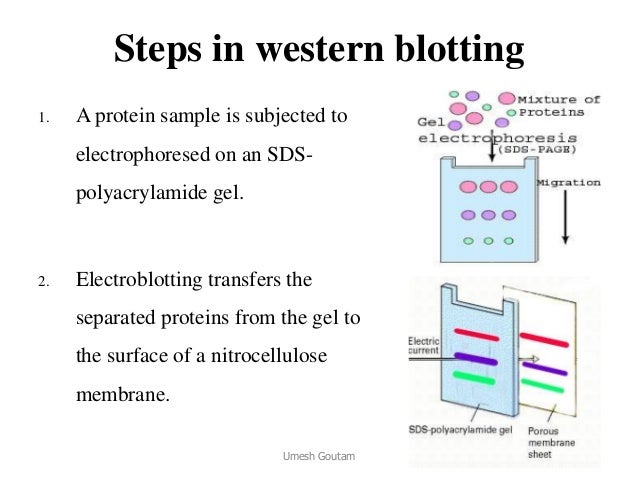
Western blotting is frequently used for the confirmatory medical diagnosis of infectious diseases such as Lyme disease, HIV infection, bovine spongiform encephalopathy (BSE), hepatitis C infection, syphilis, inflammatory muscle conditions such as myositis, and certain autoimmune disorders (e.g., paraneoplastic disease). The method was named Western blot as a play on the Southern blot technique for DNA invented by the British biologist Edwin Southern. The method was first described in 1979 by a postdoctoral fellow, Harry Towbin, in the laboratory of Julian Gordon at the Friedrich Miescher Institute for Biomedical Research (FMI) in Basel, Switzerland ( Towbin, Staehelin, & Gordon, 1979 ). 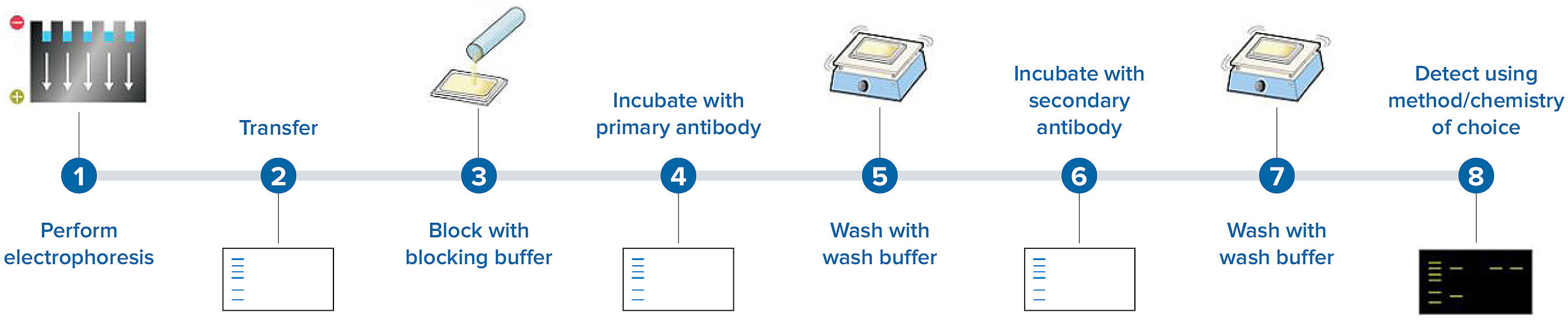
Western blotting is a popular analytical technique used in clinical analysis laboratories as well as in research ( Gavini & Parameshwaran, 2019). Slieman, Joerg Leheste, in Methods in Microbiology, 2020 4.4.1.1 Introduction Exceptions can occur when the presence of several protein antigens at similar molecular weight levels, or nonspecific antibody binding, causes false-positive test results. The specificity of Western blot results is usually high. Another problem with Western blot assays is difficulty providing quantitative data. Further reduction in sensitivity is possible if there is variability in the amount of target antigen in tissue applied to the gel, or if the antigen on the paper deteriorates with storage. 32 Western blotting may be a less sensitive antibody testing method than ELISA. In the evaluation of polyneuropathies, Western blot methods are commonly used in confirming antibody binding to myelin-associated glycoprotein (MAG), 138 tubulin, 29 and the Hu family of antigens. Western blotting is useful for detection of antibodies to protein antigens, especially to confirm the specificity of antibody binding to protein antigens detected by other methods such as ELISA or immunohistochemistry. The proteins on the paper that remain unstained provide a useful inherent control that confirms antibody specificity. A second antibody with an attached marker stains and allows visualization of the bound antibody from patient serum. If antibodies to one of the tissue proteins are present in the serum, they will bind to a band at the appropriate molecular weight region on the paper. 169 Western blot methodology involves (1) an initial separation of target tissue proteins on an electrophoretic gel according to their molecular weight, (2) transfer of the separated proteins to a strip of paper, and (3) exposure to diluted serum. Western blotting is typically used to evaluate the ability of antibodies to bind selectively to tissue proteins of known molecular weights. Before the next step (detection of bound antibody), nonbound primary antibody has to be washed out thoroughly.ĪLAN PESTRONK, GLENN LOPATE, in Peripheral Neuropathy (Fourth Edition), 2005 Western Blot Due to the thickness of the membranes, incubations and washing steps are often longer than for solid phase immunoassays on nonporous surfaces such as microtiter plates. Only a small fraction of the antibody in the incubation mixture will be bound to the immobilized antigen, even if monospecific antibodies are used. 
As a rule of thumb, solutions of 1 μg ml −1 specific antibody incubated for several hours is a good starting point for optimizations. Hence, the signal ultimately generated will depend on the interplay of numerous factors such as affinity, concentration, duration of incubation, nonspecific binding, etc. In most respects, considerations given to solid phase immunoassays in microtiter plates also apply to western blotting. Western blotting may be performed with both polyclonal and monoclonal antibodies. Harry Towbin, in Encyclopedia of Immunology (Second Edition), 1998 Probing with specific antibodies
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
